Even a bioreactor intended for simple tasks can provide a high level of capability. We show you the six must have features and how they can increase the ability to characterize cultures more thoroughly.

This post will discuss the must have features any modern bioreactor should have to do the job for most applications:
- Get metabolic information using exit gas analysis for O2 and CO2
- Continuously monitor biomass using a turbidity sensor
- Precisely control the dissolved oxygen level using mass flow controllers (MFC)
- Plan and automate batch strategies by connecting the bioreactor to a powerful bioprocess software via network
- Integrated touch screen for a dedicated local display and user interface
- Good handling and compact footprint
All of the above add substantial value to even the simplest applications. Best of all, they can be easily integrated, making for compact, reliable solutions.
1. Get metabolic information using exit gas analysis for O2 and CO2
An aerobic culture growing on a carbon-based substrate uses well-known metabolic pathways to produce energy. Oxygen is consumed, while carbon dioxide is released as a waste product. Exchange between gas bubbles in the culture liquid dissolves oxygen and allows carbon dioxide to be removed as a gas. These changes in the gas composition can be measured in the exit gas using an exit gas analyzer, effectively allowing for “metabolic monitoring”.
Knowing the O2 and CO2 concentrations also allows the calculation of further interesting metabolic parameters. The amount of O2 stripped from the gas yields the Oxygen Uptake Rate (OUR), an indicator for how much oxygen the culture used. In contrast, the Carbon Evolution Rate (CER) is an indicator for how much carbon dioxide the culture generated. The ratio of CER and OUR is called the Respiratory Quotient (RQ), an indicator for how much carbon dioxide was generated for the quantity of oxygen consumed. This should have a value close to one for normal aerobic metabolism. The expectation is that as the culture density increases, the Oxygen Uptake Rate (OUR) and the Carbon Dioxide Evolution rate (CER) of the cells increase as well. The OUR and CER values may be analyzed independently for some applications.
It is possible to calculate the RQ value in real-time using a soft sensor. This relies on knowing the inlet gas flow rate (from a mass flow value) and the values provided by the exit gas analyzer. The RQ can then be used in an advanced batch strategy to adjust the rate of a feed pump delivering substrate into a fed-batch culture.
A classic example is the efficient production of Baker’s yeast. The maximum biomass is needed without providing excess sugar, which would be converted to ethanol. If the RQ is below one, then the feed rate goes down. This allows the available dissolved oxygen to be used efficiently. If the RQ value is above one, then the feed rate can be increased.
The raw values can also be inserted into soft sensor scripts. These relate the CER to yield of biomass, growth rate or productivity, depending on the substrate used for feeding. This is particularly useful in continuous culture for metabolic studies. Having this option in a bioreactor gives you access to a key step in bioprocess monitoring and control by cell metabolism.
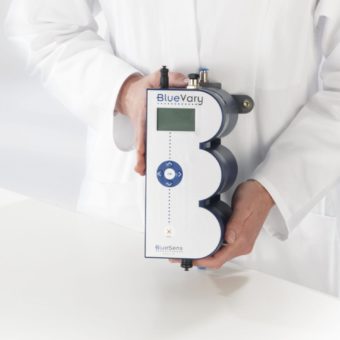
An exit gas analyser, e.g. by BlueSens is used to measure changes in the gas composition.
2. Continuously monitor biomass using a turbidity sensor
The traditional way of estimating biomass is a simple process:
- Take a sample from the bioreactor and dilute it if required
- Measure the optical density using a spectrophotometer (or light scattering)
- Record the value (on paper, spreadsheet or in bioprocess software)
- Correlate optical density information with actual biomass concentration
This method, although more accurate, has some issues e.g. important characteristics of the culture’s metabolism will be missed between sample intervals, which is counterproductive for real-time monitoring and control.
While a turbidity sensor does neither measure the optical density nor the biomass, its signal can be linearized and correlated with both. Having a sensor inside the vessel offers many advantages:
- Measurement is in real-time. No more guessing what happened between samples
- Values are recorded locally at the bioreactor and in real-time using bioprocess software
- The value can be used for batch phase transitions or for control e.g. of a harvest pump for turbidostat operation.
Turbidity sensors are not new. Sensors exist for small, bench-scale bioreactors and can be integrated directly into the local controller.

Optek turbidity sensors can be used to estimate biomass in real-time
3. Precisely control the dissolved oxygen level using mass flow controllers (MFC)
With the exception of anaerobic cultures, a precise control of the dissolved oxygen concentration is often vital for the success of the bioprocess. The gassing system plays an important role in this process, as it not only controls the flow rate, but ideally also the composition of the gas entering the bioreactor.
Long gone are the days of rotameters that needed manual intervention for flow rate changes. Gas mix systems for air, N2 and O2, however, are still often based on a pulse width modulation (PWM) based switching of valves connecting the individual gas supply lines. These often exhibit a lower stability of the flow rate and, most importantly, a pulsed supply of the individual gases into the bioreactor which can limit the performance of the bioprocess.
A gas mix system based on one MFC per gas outperforms other solutions with ease. Air, N2 and O2 are blended precisely before even entering the culture vessel, and the flow rate is always kept at setpoint. This results in a more stable dissolved oxygen concentration and, in consequence, a more performant bioprocess – even when running high cell density processes with oxygen supplementation.
Additionally, a multi mass flow controller system can accurately provide information on the inlet gas mixture, which is essential for precise calculations of CER, OUR, and RQ (see section 1 above).

Mass Flow Controllers – here an example of Vögtlin – are a conventient tool measure the gassinig mixture.
4. Plan and automate batch strategies by connecting the bioreactor to a powerful bioprocess software via network
The rise of potent bioprocess software has empowered bioreactors even more and allows the user to automate their batch strategies with ease from a centralized system. Therefore, a secure, comprehensive protocol for transferring data over a network is now obligatory for almost any scientific or industrial equipment to enable:
- Recording of process data
- Remote starting and stopping of batches
- Sample taking shown automatically and incremented in bioprocess software
- Changes to the process control for each parameter
- Changes to alarm limits
One question, however, remains: how to connect to a bioprocess platform software? One standardized solution has emerged from the jungle of network protocols: OPC UA. After being around for decades as OPC DA and OPC XML DA, the latest incarnation of the protocol is OPC UA (Universal Architecture). This is standard in process automation and one of the technologies powering the Internet of Things (IoT). It is flexible, robust and easy to interface. It is widely accepted and, as a network protocol, can work across company networks and the internet if required.
Users can connect bioreactors with an open, standardized protocol into modern bioprocess software by the click of a button while complex driver development may be required for proprietary protocols. Modern bioreactors should not be limited to offline use with only local data recording and limited process automation capabilities. Two-way communication with a bioprocess software enables the use of automated batch strategies, control workflows and soft sensors to supply an extra layer of sophistication.

Network connectivity to bioprocess software via OPC UA.
5. Integrated touch screen for a dedicated local display and user interface
A bioreactor with integrated HMI (Human Machine Interface) is state-of-the-art nowadays. This is typically a color touch screen, so it has the same size and functions of a tablet. This is critical for long-term security of supply, support and maintenance.
Advantages:
- A link to an Ethernet connection is provided for reliable network communications
- The HMI is dedicated to the bioreactor itself and cannot be used for ancillary tasks
- A USB port can connect to a memory stick recording data or providing updates. It also allows the use of a Wi-Fi dongle if a wired solution is not possible.
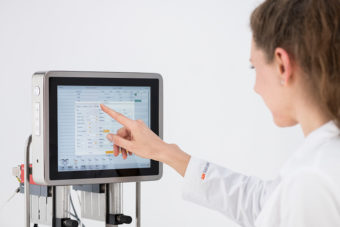
A integrated touch screen uses Ethernet and USB ports for data transfer.
6. Why a good handling is important when working with a bioreactor
The attenning cords, cables, tubing and connectors that come with bioreactors are intimidating to many who are just starting out. Some of these are unavoidable, but it is possible to find systems that have been optimized for easier use, specifically:
- Use of a thermal block for heating and cooling can offer easier handling, either for inexperienced users or when space is restricted.
- The vessel, reagent bottles and tubing all need autoclaving. Components mounted on a single framework and taken as one piece makes handling easier and safer.
- Removable, autoclavable pump heads make sense if everything is on one frame for autoclaving. This allows the user to leave all components connected to the headplate, minimizing the possibility of contamination after sterilization

Summary:
Even a bioreactor intended for simple tasks can provide a high level of capability. All the baseline requirements reviewed in this post increase the ability to characterize cultures more thoroughly while automating the process.
The key points to consider are:
- An exit gas analyzer for metabolic control
- Turbidity measurement as real-time indicator for biomass
- Mass flow controllers for perfect gas blending and precise dissolved oxygen control
- Use of a modern OPC protocol for network-connected data transfer.
- An integral touch screen for a dedicated approach to HMI
- Several ways to simplify handling during everyday bioreactor use.

Shaman Gaspar
Dr. Allman, couldn't have been any better said. Precise, concise and reveals all that is necessary to summarise the brilliance of the Minifors2 and eve.
Pamela Fuentes
Congratulations!!!!
:)