The scale–up from bench-top bioreactors to larger, pilot systems can seem daunting. The list of user requirements specifications is much longer, and the manufacturer will provide a peculiar drawing called a P&ID. Reading these is second nature to a Bioprocess Engineer. For someone without such a background, this may be a strange concept. Nevertheless, with a bioreactor in the centre of your process, it is vital to link bioprocess technology and mechanical engineering in a schematic representation. This article shows you how to interpret your P&ID and to understand the functionalities of your bioreactor and its capabilities.

1. What is a P&ID?
Definition, Characteristics & Difference to Process Flow Diagrams
A Piping and Instrumentation Diagram (P&ID) illustrates pipelines, equipment, instrumentation, and control systems found in process environments such as Chemical or Biotechnological Plants. Playing a big role in process engineering, such detailed schematic representations form the basis of communication for all parties involved in the planning, operation, and maintenance of the plant.
P&IDs are not necessarily drawn to scale or geometrically accurate nor do they relate to the actual location of the components. They simply form a schematic illustration of the functional relationship of piping, actuators and controllers, using a standard layout and set of symbols.
Nevertheless, having access to a P&ID indicating tag numbers on valves (often in a simpler form on the Controller) enables you to intervene in the process in cases of urgency.
A P&ID does NOT provide any process data – that is the domain of a Process Flow Diagram (PFD), which highlights the primary process flow, without any details about minor components or piping systems. Basically, it can be used as a first step to define the user requirements specifications (URS), which will be implemented in the P&ID by the manufacturer.

A Piping and Instrumentation Diagram (P&ID) illustrates pipelines, equipment, instrumentation, and control systems. It forms the basis of communication for all parties involved in the planning, operation, and maintenance of the plant.
2. What are the P&ID Symbols?
A wide range of applications means a variety of symbols – common procedures & support.
Due to its wide usage in process environments like Oil Refineries and Chemical or Biotechnological Plants a wide range of symbols and schemes exists. The instrumentation symbols follow a standard set by the International Society of Automation (ISA), the ANSI/ISA’s S5.1-1984 (R 1992). However, according to EN ISO 10628, a user can adapt and supplement graphic symbols and add to them if he does not consider them clear enough. Thereby, the readability of the scheme can be improved. Detailed legends are essential in helping you to find your way through a P&ID. This is especially important when using symbols that have been customized.
“Each customer process is unique. To meet these challenges, manufacturers often use specific symbols based on the standards. Clear identification is crucial – detailed legends in the P&ID provide a good basis for understanding.”
B. Fanti Project Manger Custom Engineering INFORS HT
Whether according to the standard or only based on it, the creation of such P&IDs is also determined by the requirements and expertise of the creator. Software applications designed for this purpose, such as AutoCAD, are often the first choice in mechanical and plant engineering and offer additional platforms for support.
Working with these applications often requires appropriate training and experience. For the quick creation of P&IDs without the necessary years of experience in CAD drawing, simpler programs such as Visio from Microsoft are ideal. These also facilitate the easy creation of simple P&ID’s with a variety of add-ons.
Despite the clear identification of the symbols with legends or comprehensive support by software applications, a basic knowledge of the frequently used symbols such as lines, valves and actuators or identification letters for process control systems is advisable. There is already a lot of explanatory material available.
Let us know if you would also like to have our symbol overview provided by commenting on this post or writing to your sales representative.

Software applications such as AutoCAD are often the first choice for Bioreactor Design. When using customized symbols in a P&ID clear identification is crucial – Ask for a corporate overview of the most commonly used symbols.
3. How to create a P&ID based on my bioprocess?
Relating the bioreactor capability to the wider bioprocess.
The outline of the bioprocess forms the basis for key elements of the bioreactor design. Whether a fed-batch with a two-step strategy, as is typical in industry or a chemostat for continuous production, the design of the plant is determined by the desired bioprocess.
If for instance, a fed-batch with growth and production phases is to be achieved, these requirements must be considered in the bioreactor design and thus the creation of the URS. For feed control, the correct equipment such as pumps and, depending on the feed type (gravimetric or volumetric), load cells for weight-controlled addition must be planned and included in the P&ID.
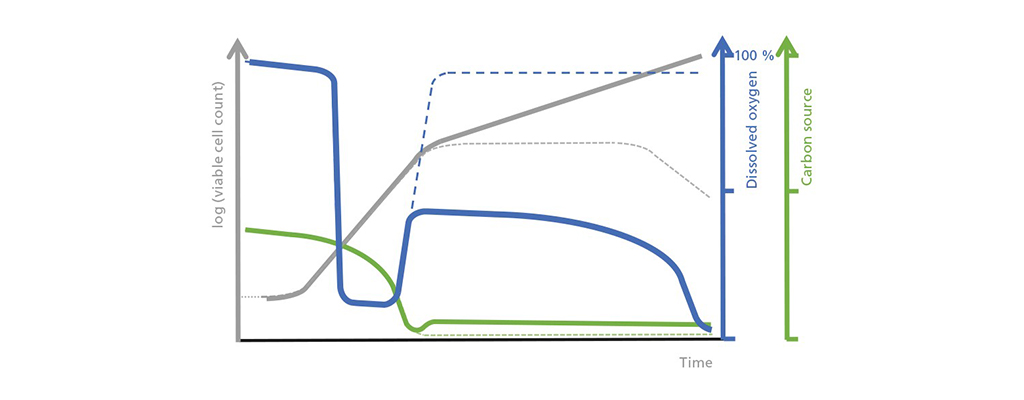
Schematic illustration of the relationship between the living cell concentration, dissolved oxygen, and the limiting carbon source in the fed-batch process. When implementing a fed-batch process, you will need to adding the feed immediately after the exponential phase, to prevent the carbon source from being exhausted (thick green line vs. dashed green line). Shown here is an exponential feeding process in which exponentially growing organisms remain in a prolonged exponential phase (thick gray line vs. dashed gray line). This also means that the quantity of consumed oxygen increases, which is why the amount of dissolved oxygen in the medium is lower (thick blue line vs. dashed blue line). In this example, a growth rate μ less than μmax has been selected for the feeding phase. A fundamental understanding of the changes in the fed-batch mode is a requirement for a reliable bioreactor design & subsequent creation of the P&ID.
While control over the supply, discharge, and homogeneous distribution of mass (medium, nutrients, gas etc.) is crucial, heat distribution must also be considered in the bioreactor design. Here, too, the bioprocess strategy plays a decisive role; in exothermic cultivations such as many microbial processes, a fast-growing, high-density culture will generate a lot of excess heat. The required cooling capacity must be ensured by the appropriate supply and discharge in the system.
Homogeneous mixing is compulsory for successful cultivation. Naturally, homogeneous distribution of all components is important, but the transfer of oxygen from the gas phase to the liquid phase still presents a major challenge in the cultivation of microorganisms.
In high-cell-density cultivations, the measured dissolved oxygen concentration (commonly referred to as DO or pO2) may fall below the critical value. Increasing the Oxygen Transfer Rate is accomplished by adjusting parameters such as head pressure, air supply rate, agitation speed and oxygen supply rate. With the P&ID, you should be able to see quickly whether the technical requirements, such as pressure and gassing rate, allow an adjustment to be made.
A cascaded control strategy is commonly utilized, in which the above parameters are changed in dependence on each other. The use of oxygen-enriched air can also be considered, but with added costs to the process.
When conducting the bioprocess, having a P&ID close to the bioreactor (often in a simple variation on the Process Controller) is crucial for a quick overview as well as for troubleshooting in the case of an unexpected event.
Depending on the process control system, you might have to interact with the P&ID via Process Controller for even basic steps like starting a feed. The trend is towards using Software Platform Solutions that enable remote control & process automation.
4. How do I read a P&ID ? – Case Study: Techfors TV 100L
How the multiple lines and symbols can relate to the cultivation strategy
Experience shows that the best way to learn is by example. This is a current bioreactor project at the Norwegian University of Life Science (NMBU), Faculty of Chemistry, Biotechnology and Food Science.
There are different approaches to interpreting a P&ID. While the Biotechnologist starts with the bioreactor at the heart of the process, an engineer likes to orientate himself based on the supplies provided.
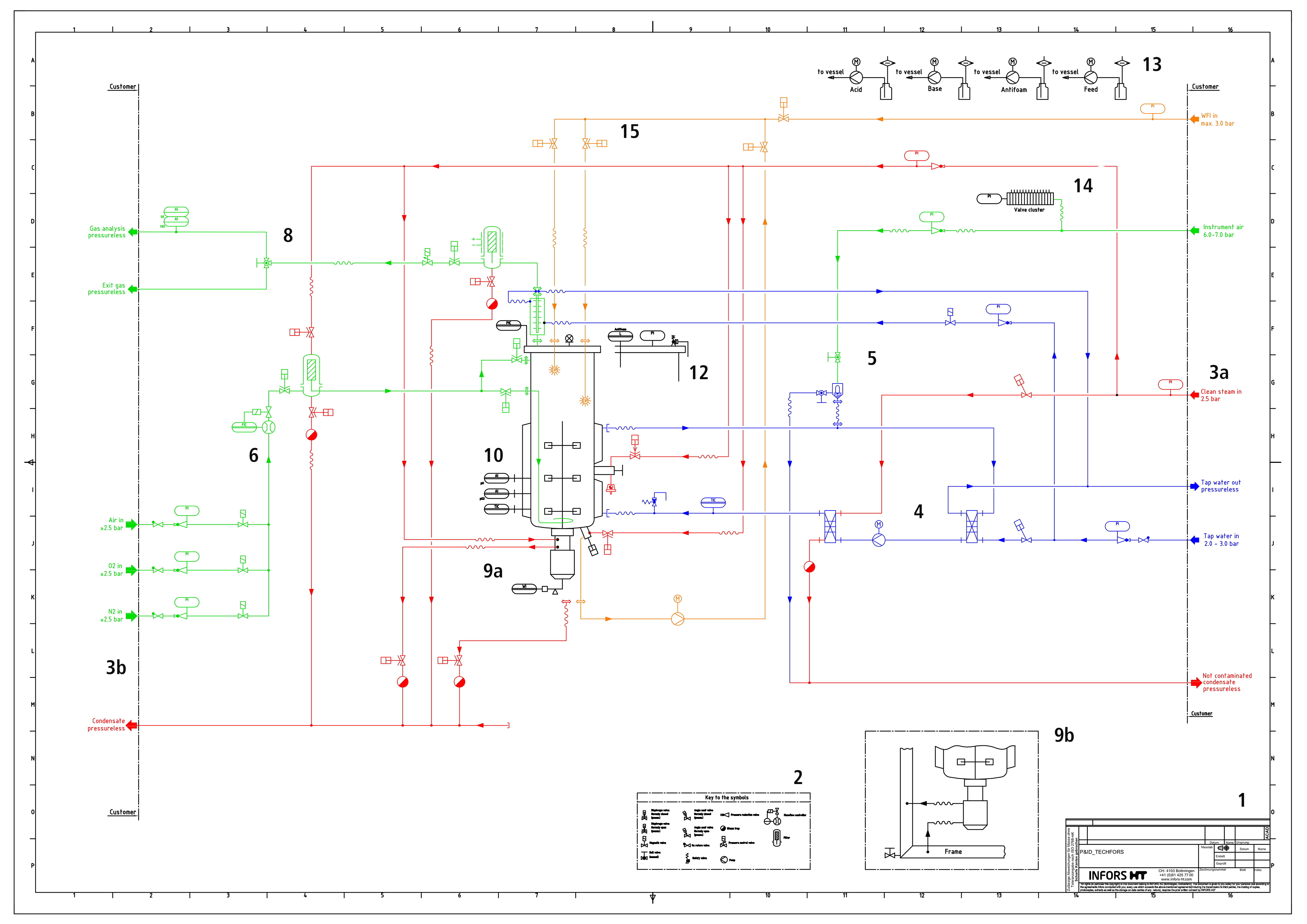
1) A drawing number will relate the P&ID to other information such as dimension drawings for a specific project or type of bioreactor. 2) Key list of symbols used. 3) Dividing line between Infors equipment and for the system required energies which are the customer’s responsibility to provide. 3a) Water Supply for cooling & heating (blue), CIP system (yellow) as well as instrumental air (blue) for pneumatic valves & expansion vessel. 3b) Inlet & exit gas. 4) Closed temperature control circuit with plate heat exchanger (hot, connected to clean steam) and 01.21.02 (cold connected to tap water). 5) Reservoir for closed circulation system for temperature control (expansion vessel). By manually adding compressed air (via the manual ball valve), the expansion vessel can be emptied. 6) Mass Flow Controller for the combined to a Gas Mix inlet gas supply of Air, O2 & N2 (green). 7) Exit Gas (green) with cooler and filter. 8) Manual ball valve to analyse exit gas components as O2 and CO2 (green) if required. 9) Motor with 9a) electrical load with control function, RMP sensor and weight sensor. The supplied steam is used to lubricate the mechanical seal. 9b) The coolant for the motor is contained in the steel frame of the equipment. 10) Sensors to control of pH, dissolved oxygen (pO2) and temperature. 11) Sampling valve with condensate outlet. Flexible connections like tubing for clean steam supply shown as wavy lines (used for sterilization of the sampling valve. The pneumatic diaphragm valve is normally closed. Since it is not for manual operation, a sequence for sampling needs to be automated and available in the Process Controller). 12) Safety relief valve. 13) Peripheral items such as reagent bottles for Acid, Base, Antifoam and Feed. 14) Control elements such as a “valve island” for control of pneumatic valve with instrument air. 15) Cleaning in Place (CIP)
Essential elements like the drawing number (1) and symbol legend (2) give an initial orientation.
On the right side (3a) the services to be provided by the customer are shown, such as:
- cold water for cooling
- steam for heating the temperature control system (4, blue)
- instrument air (blue) for operating the pneumatic valves (14) as well as for controlling the expansion vessel (5)
- Water For Injection (WFI) for the Cleaning in Place (CIP) system (15, yellow)
In this case, the water used for temperature control (Tap water out) can be discharged without any problems. The situation is more complicated for systems in a closed internal water circulation system or in a laboratory without wastewater drainage; consequently, pressure and temperature values must be considered and corrected if necessary.
On the left side (3b) you will find:
- the waste condensate
- effluent removal
- analysis of process-relevant gases (green); air, oxygen and nitrogen are controlled as a gas mix by one mass flow controllers (MFC, 5)
Thus, although the gases cannot be controlled individually, the gas mix can be controlled by an appropriate regulation (-100% for N2, O% for Air and 100% for O2). This must be taken into account in the case of a two-point calibration of dissolved oxygen (pO2) with N2 and O2. Filtered by the supply air filter, the gas mix is fed through the pneumatic block diaphragm valve into the bioreactor through the sparger. Disrupted into small bubbles by Rushton stirrer, the gas mix is converted in the microbial process and the resulting metabolic components are led away from the bioreactor. The exhaust cooler reduces the temperature of the exit gas to avoid losses of liquid volume caused by evaporation due to moisture saturation. Exhaust gas filtering (7) is followed by exhaust gas analysis of O2 and CO2, which may allow the use of soft sensors.
As the heart of the plant, the 100L total volume bioreactor has a bottom-driven Rushton agitator, usually used in microbial processes. The motor (9a) is controlled by an electronic module with a regulating function and cooled by the coolant contained in the frame of the system (9b). Below the reactor is a load cell, which is typically used for calculation and monitoring in gravimetric feeding.
In addition to the conventional process controls such as pH, pO2 and temperature (10), there is a side sampling valve with a condensate outlet (6), which can be controlled by a pneumatic diaphragm valve (normally closed) through an automated sequence.
In addition to the anti-foam probe and the pressure gauge, the head plate also features a safety device in the form of a safety relief valve (12). This is an intended breaking point that protects both the equipment and its users from dangerous overpressure or vacuum in the system.
Ports for the reagents (13) as acid, base, antifoam and feed, are also located at the top of the head plate. It is notable that no additional feed pump has been planned. This means that with a 2-step feed strategy, either Feed 1 must be removed or the pump for Antifoam must be reprogrammed and used. Depending on the process and the necessity for using chemical foam dispersal, the reagent can be added manually by pulses.
After harvesting, cleaning of the bioreactor and all associated transfer lines follows.
Cleaning in place (CIP) removes culture residues from the process equipment and transfer lines. This is achieved by spraying and rinsing water and cleaning solutions in circulation, without the need for removal or disassembly (15). The cleaning scheme uses a pre-rinse to remove larger contaminants, while a hot caustic rinse disposes of any remaining contaminants. Lastly, a water wash removes caustic residues, and water for injection (WFI) cleaning finalizes the cleaning process.
5. What’s next?
A summary of this overview and how to find out even more in a follow-up interview.
P&ID’s applications are as unique as the process itself. Whether the focus is only on a small aspect such as the bioreactor in the upstream process of a biotechnological production, or the complete process from the initial media fill to bottling the finished product, everything is represented schematically. What is important is the basic understanding of its use and the best possible orientation within it. From reading the P&ID, details of a process can only be inferred.
The key points covered are:
- The definition of a P&ID for a bioreactor
- Its competent symbols and ways of illustrating the hardware systems present
- What it shows the user and the ways in which that knowledge is useful
- The links to aspects of the wider bioprocess
In the next blog post, the process design from the customer’s perspective will be looked at more closely.
Has your interest been sparked & you want to know more? Write in the comments what questions you want NMBU to answer about their new Techfors.

Sree Madhavi
"Thank you for sharing this insightful blog post about P&ID and its application in understanding bioreactors. P&ID diagrams are invaluable tools for comprehending the bioreactor's processes and components. They provide a visual representation that aids in troubleshooting, optimizing operations, and ensuring efficient bioreactor performance. Great explanation!"
JyothiSree Madhavi
Thank you for sharing this informative blog post about P&IDs and their relevance to understanding bioreactors. A P&ID (Piping and Instrumentation Diagram) is a visual representation of a system's piping, instrumentation, and control devices. It can help you comprehend the bioreactor's intricate processes, identify components, and analyze their interactions. Valuable information, indeed!
Bridgemed Solutions, Inc.
I loved the post, keep posting interesting posts.
MARCOS RIZZO
Quero saber sobre Produtos Infors
Valentina Haag
Dear Marcos, thanks for your interest & comment - much appreciated.
The easiest way to get information about our products is through our Local Business Partners, which you can find following the link below;
https://www.infors-ht.com/en/contact/
Brian Osterwalder
Very detailed!
Luis Ramiro Canales
Very interesting
Yogin Patel
NA