In today’s world, bioprocess software solutions are an essential tool for modern laboratories. Bioprocesses are controlled using process data and optimized with the help of mathematical models based on the gathered data. Manual experiment planning and data documentation cannot keep up with this in the long term. Inefficiency, duplication of work, a lack of planning and poor or inconsistent data quality can have devastating consequences. If you want to have the quality, cost, and time-to-market of your products under control, it is essential to consistently digitize your bioprocesses – starting with the preculture in the incubator shaker.

Modern bioprocess platform solutions support the entire bioprocess development from beginning to end, including:
- Experiment planning and recording of units in use
- Keeping track of who is using the equipment
- Collecting, analyzing and visualizing data from different sources
- Sharing data with a range of users
- Reducing time spent on simple control tasks
- Allowing remote access to incubator shakers
1. Experiment planning and recording of shaker units in use
As incubator shakers are often a shared resource, forward planning makes life easier for all users involved. With the use of a bioprocess platform software, different users or groups can plan experiments in advance, check which equipment is available and allocate it according to their needs. The total number of planned experiments and units required will give a lab manager an idea of the total workload. This could also allow for a combination of experiments to run simultaneously in units with the same temperature and shaking requirements.
Once in use, lab managers can easily check which units are active, who is using them and for what purpose and length. A text logbook capability lets users leave messages about any issues, observations or changes. This allows for a centrally controlled re-allocation of resources, if e.g. service work is needed on a specific unit.
Add up all the above and it’s easy to see where better organisation can save resources.
2. Keeping track of who is using the equipment
If an incubator shaker is part of a seed train for a validated product, it will need the same degree of supervision for regulatory processes. This means strict user management and a full audit trail should be available. In some cases, small quantities of a new product may be produced in the incubator shaker for use in clinical trials. With an intelligent bioprocess platform, even the Food and Drug Administration (FDA) initiative regarding Process Analytic Technology (PAT) and data from microwell plate culture could be included in a license submission.
The trend to continually increase yield of product in terms of g l-1 makes the use of fully qualified incubators increasingly likely. Apart from the testing of the equipment, the need for qualified bioprocess software to go with it is clear.
Two key aspects are:
- User Management:
It is vital to give access only to authorized users, who can be uniquely identified. Other aspects of user management include forcing the use of strong passwords, password-aging and inactivation of users when required. Different access levels are necessary, to allow e.g. a regulator to review archived data without being able to influence a running process.
- Audit Trail:
This should record every significant action during a running process, with a time stamp and a specific user assigned. Administrative processes and changes should also be recorded. While this level of detail may seem unimportant for most incubator shaker users, it can be of value in a validated environment.

3. Collecting, analyzing and visualizing data from different sources
It is an important but sometimes tedious task to visualize all the data gathered from different sources in order to get an overview of an experiment. Often, this is done by copying and pasting data from one format into another, or from one excel sheet into another. In terms of data integrity this approach is quite a risky one, as every failure in the process will have an impact on the raw data generated during the experiment. Even if all the process data is in place and merged to one data format correctly, the information regarding culture media and specific organisms may be stored in a different location. Therefore, a bioprocess platform software can integrate many different devices (e.g.: analytical devices or process devices from different suppliers) and store all process data and information in one central database. If there is a change in personnel, the software guarantees you a seamless data transfer from one employee to another.
A common data structure avoids the merging process and allows for direct visualization of the gathered data in one plot. Speaking in terms of incubator shakers, this allows for a clear comparison between one unit and another, the individual decks of the shaker, or last week’s experiment with this week. The possibility to compare historic with current data can be used to optimise your shake flask processes.
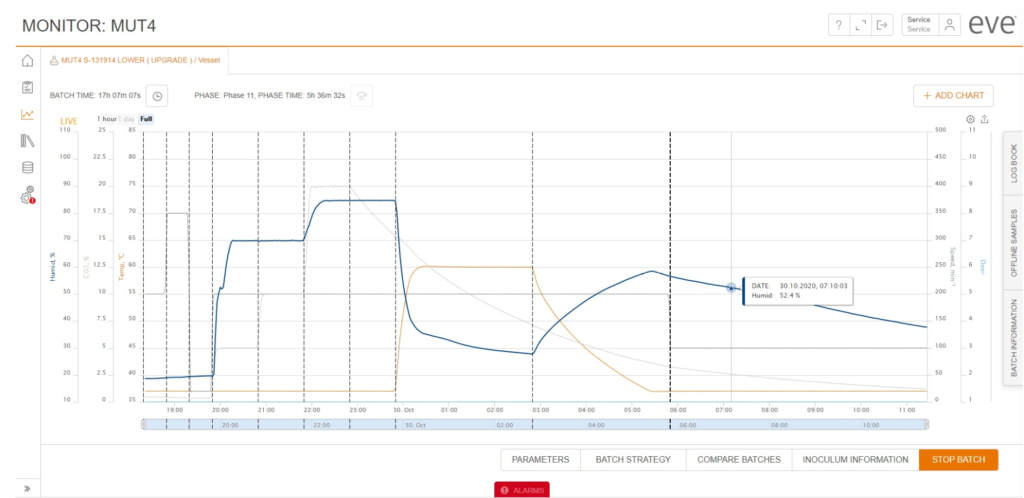
Chart overview of incubation shaker data using the eve bioprocess platform software with parameters temperature, humidity, shaker speed, door status (open/closed) and process phase changes. In addition batch and phase time as well as current alarms are displayed.
4. Sharing data with a range of users:
The days when research was separated from other departments have long gone. A bioprocess now attracts many interested parties. This comes down the line to growth in incubator shakers. The reasons for a wide range of users having controlled access to experimental data include:
• Quality Control
For seed production, examination of many repeated batches can provide insights for Statistical Process Control (SPC) or Quality by Design (QbD) purposes. For example, if a small change in temperature could improve yields, this could be investigated in shake flasks.
• Production
If the shake flask cultures are to be the seed for inoculation of bioreactors, then the data concerning optimum time for harvest and cell densities is vital. If direct monitoring of biomass can take place in the shake flasks, this would be the ideal. The values can be stored within the software containing the main bioprocess batch data.
• Other groups/departments
Having access to data from a set of experiments could be influential for improving results in an unrelated process. This can be in a “guest mode” at the simplest level, where it’s possible to look but not make any changes. This clearly works best where several groups use the same bioprocess software on a common server.

Remote data analyzing in a modern laboratory.
5. Reducing time spent on simple control tasks
Automated control of incubator shakers can have a big impact on both the bioprocess itself and the lab resources needed.
Examples include:
- Automated temperature shifts to enhance growth or induce production of a specific biochemical i.e. a step change.
- Reducing the temperature after a productive phase to preserve a product or suspend cell growth i.e. an end phase.
- Cyclic processes, including repeated changes to temperature or speed for fixed times i.e. repeated control loops.
- A change to a specific parameter at a key time e.g. turning off CO2 control in cell culture when logarithmic growth begins i.e. response to a metabolic change.
- Link online measured cell density (e.g.: backscatter measurement) to shaking speed and control the oxygen transfer into the culture.
- Calculate online values for concentrations and metabolic rates by means of soft-sensors. These values can then be used for automated process control. If you’d like to find out more about soft sensors and their use for bioprocess control have a look at: How Soft-Sensors Can Help You Unlock the Full Potential of Bioprocess Data
- Triggering the start of a feeding; either time dependent or cell density based.
Critically, it removes the possibility of an important step being forgotten or someone having to come into the laboratory out of hours to make a change manually. The fact such changes are logged and alarms available can prevent issues even if the set point control function is not used.
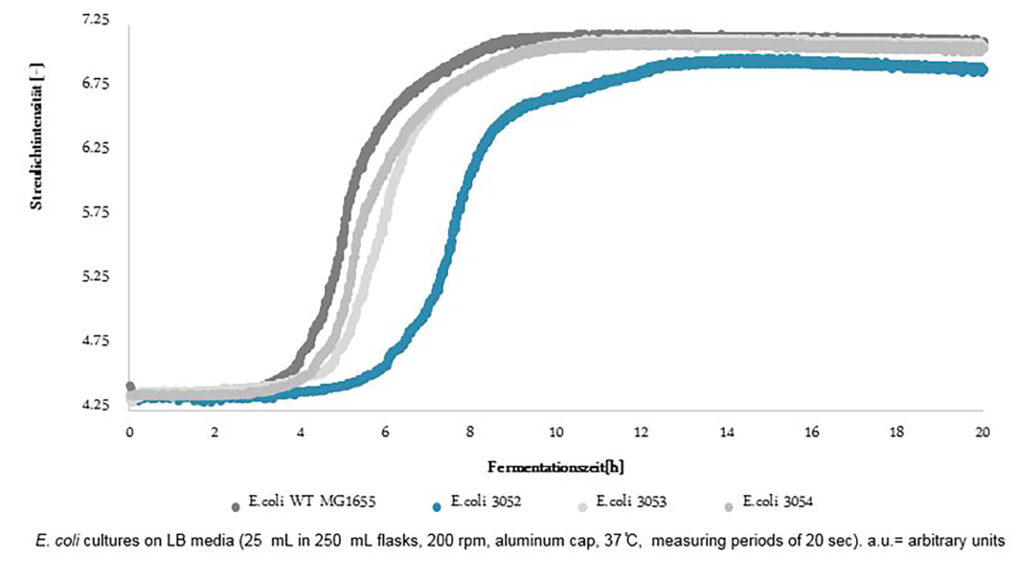
Measurement of scattered light intensity overtime using the Cell Groth Quantifier (CGQ) in an incubator shaker.
6. Allowing remote access to incubator shakers
A big advantage for departments spread over several floors in a building, is remote access for any user via a web browser. Experiment planning, monitoring and data analysis can be performed in the office – or even from home. Also, a manager can see if a unit is active that has not been assigned as part of any planned work. Without having to physically find the unit and identify the user, adjustments to resource planning can be made.
Alarm monitoring is a vital function for many users. A local alarm is good for times when the laboratory is staffed but any events that occur out of hours would go unrecorded. The only remaining evidence of a problem might be a poorly growing culture. Sending alarms by email covers out of hours events and a decision can be made about how to proceed at the time the problem occurs.
Summary
This artice presents a case for typical incubator shaker users to consider the advantages of linking their equipment to bioprocess software. Not every argument will apply in all cases but each one that does carries a potential saving in time, costs and / or labour.
- The case is strongest for a validated process subject to regulatory monitoring, where user management and an audit trail are required.
- Research groups can also benefit in terms of allowing different levels of people-controlled access to data which may be indirectly useful.
- Academic and institutional users benefit from centralising information about separate units spread over several locations and their use.
- Individual users can benefit from trend graphs, comparison, control and alarm monitoring functions which bioprocess control software can achieve.
With different versions and purchase models of bioprocess platform software, it should be possible to discover and try out the unique fit of features for your application. One location and data structure for your results with easy access by several users will appeal to many. The objective to reduce work and optimize resources are beneficial to everyone. Incubator shakers may be simple, but their uses aren’t.


Stanley Lambert
Ce site est intéressant
Kasireddy Venkatreddy
9177557735
Rsj company
Nice collection, thanks for sharing this blog.