Mammalian cell culture bioprocesses are increasingly gaining importance in the biopharmaceutical industry. Due to the high demand of product quality, the incubated shaker as a production unit can become a limiting factor after a certain point. Learn more and watch the webinar at the end of this post.

The typical process cycle
A specific product is usually produced by thawing the cells, cultivating these up to a certain amount of cell density in incubated shakers, followed by then transferring the process into a bioreactor. To increase the amount of product, the bioprocess is accordingly finally scaled-up to different unit sizes. However, such a sequence of steps is easier said than done. Many different factors, biological as well as physical parameters, need to be considered for a successful process transfer. Therefore, this post is aimed at shedding light on these.
Get to know your bioprocess using real-time monitoring

The selection of cell culture cultivation units always depends on the application. Shake flasks are easy to handle, disposable, and allow quick preparation for performing bioprocesses. The shake flasks are often used for screening purposes or to understand the process in its initial stages. On the other hand, for a detailed understanding of the process, it is essential to gain insights into the bioprocess through real-time monitoring and control. Shakers allow this only to a limited extent, in which the information relevant to the process remains an unknown black box. This is because it can take up to hours to several days to analyse a sample and obtain the corresponding data.
A tight real-time control is needed not only to identify the Critical Process Parameters (CPPs), which are important to be known to control the biology of the cells, but also to meet the regulation criteria regarding quality, efficacy and cost effectiveness (Asnaghi, Smith, Martin, & Wendt, 2014). To fulfil these monitoring requirements, it is crucial to have an automated bioreactor system which prioritizes real-time control, monitoring, data recording and management by employing a software that performs exactly those functions.
Be aware of critical factors for process transfer
A process transfer combined with a scale-up step from shake flask to bioreactor is not always easy and smooth. The environment for the cells changes totally, starting from the aeration to the cultivation method itself. The environment for the cells changes totally, starting from the aeration to the mixing method itself. The fact that the cells are not being shaken anymore and rather being stirred results in a change of mixing within the bioreactor. This dramatic change for the sensitive mammalian cells leads to shear stress and foaming issues, but nowadays these are quite well controlled due to the addition of antifoam and cell surfactants (Xing, Kenty, Li, & Lee, 2009).
Besides the observations mentioned above, few more critical engineering parameters exist that need to be considered:
1. Oxygen transfer:
Since mammalian cell culture processes are aerobic, the oxygen transfer into the cells is a very critical factor. To ensure adequate oxygen availability, the agitation speed, gas flow rate and gas composition need to be controlled. As mammalian cells do not have a cell wall, the power inputs may need to be kept low, leading to inhomogeneities within the culture (Croughan, Hamel, & Wang, 1987).
2. Culture media mixing:
Another important influential parameter is the mixing within the cell culture broth which needs to be considered. Hence, insufficient mixing results in pH and nutrient gradients, which has been shown to dramatically influence cell growth and antibody production (Bylund, Collet, Enfors, & Larsson, 1998; Osman, Birch, & Varley, 2001).
3. CO2 accumulation:
Due to the low power input and aeration rates, the CO2 concentration in the culture media is elevated, which negatively impacts product quality as well as cell growth and protein production (Gray, Chen, Howarth, Inlow, & Maiorella, 1996).

Choose the right impeller design

Within every bioprocess lie a few challenges, but it is possible to overcome these. Fortunately, there is a common denominator underlying all the critical factors mentioned above – the impeller design. It helps to mitigate the negative effects by dispersing gas bubbles, leading to a better oxygen transfer and CO2 removal, which in turn leads to a more homogenous culture. However, at the same time, the impeller can play a large role in shear stress for the cells, putting the operator in a real dilemma.
One key aspect to be on the lookout for is the size of the eddies generated in the vicinity of impeller. The closer the eddy size to the size of the cells the more likely it is to observe cell damage. Optimized bioreactor designs therefore include an impeller that can operate at larger power inputs while maintaining a sufficiently large eddy size, effectively allowing for better gas transfer, mixing and CO2 removal without damaging the cell and expanding the operational window of the bioreactor.
It is a given that every modern cell culture bioreactor should provide a multi mass flow controller gassing system allowing for precise flow control and gas blending or a pH control loop that can utilize CO2 instead of a liquid acid. However, discussion of these features is not under the purview of this post.
Learn from best practices
Even though process transfer has been around in the scientific community for quite a while, the lack of expertise on the same is astounding. Consequently, a well-known phenomenon remains often underutilized or unperfected by the industry. Therefore, it can sometimes be confusing to those who already employ this technique and particularly challenging for those who are new to the field, wanting to transfer a process from shake flask to bioreactor. Notwithstanding your current level of ease with the process transfer, if you need guidance or are interested in finding out how others successfully proceed about it, have a look at the illustrative application of a characterised bioprocess transfer: “Process transfer of CHO cultivations using the Minifors 2 as an example”, by the Zurich University of Applied Sciences. The application note is available for download on the Minifors 2 product page.
The scaling-up recipe
Even today, scale-up is an erratic procedure since bioprocesses are not properly monitored and understood in detail. Two very important process relevant parameters are agitation and aeration. On the one hand, agitation is set to achieve homogenous mixing and oxygen transfer into the cells. Aeration, on the other hand, is regulated to supply the cells with sufficient amount of oxygen as well as to eliminate excess amount of CO2. Especially in large-scale bioreactors, excess CO2 concentration can pose a serious problem. Therefore, it is important to consider the aeration and agitation rates, position of the impeller and sparger, which will affect bubble size (Gray et al., 1996). The specific engineering parameters which are needed to successfully execute the scale-up process are discussed below at length (derived from DECHEMA, Society of Chemical Engineering and Biotechnology).
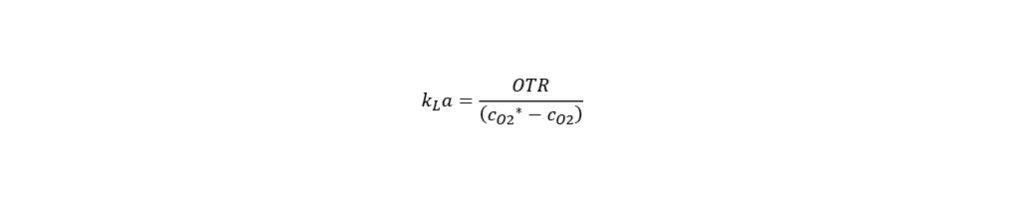
1. kLa – The volumetric mass transfer coefficient
The kLa coefficient denotes the efficiency of gas transfer from the gas phase into the liquid phase. It is dependent upon the design and operating conditions of the bioreactor. Moreover, it is also influenced by the impeller design, aeration and agitation rates. The higher the kLa value, the greater the aeration performance of the cultivation system. Several methods exist to determine this coefficient with a corresponding dissolved oxygen probe. Some of the most prominent approaches are as follows.
a) Dynamic gassing out technique
In the initial step, the bioreactor system is in a stationary state, which is disrupted by a sudden stop of oxygen supply. This causes a decrease in dissolved oxygen concentration through its consumption by the cells. After the oxygen is consumed, the supply is switched on again and the gas returns to its original .
b) Static gassing out technique
In this technique, the oxygen is gassed out of the liquid after addition of nitrogen. Afterwards, the liquid is stirred and aerated again, and the increase of dissolved oxygen is monitored.
c) Sulphite technique
This method is based on the reaction of sodium sulphite with oxygen, where the sulphite is oxidised with the help of metal ions to sodium sulphate. The consumption of oxygen then determined.
For the determination of the kLa coefficient (h-1), besides the oxygen transfer rate, OTR (mg O2*L-1*h-1), the equilibrium oxygen concentration, cO2* (mg/L) and the dissolved oxygen concentration cO2 (mg/L) need to be known.
2. Power input
This engineering parameter is linked with hydrodynamic stress, which influences the growth of the cells and their productivity, particularly those of the more shear sensitive organisms. The most prominent technique to determine the power input is the torque-based measurement. This method is very easy to apply and is therefore the most frequently used. By using a torque sensor, the torque is measured on the stirrer shaft which then allows to determine power input. It must be pointed out that it is important to provide a vibration-free environment measurement. The formula below shows how the power input, P (W) can be calculated by determining the rotational agitator speed, N (min-1) and the torque in the liquid (N*m), as well as the torque in the empty vessel (N*m).
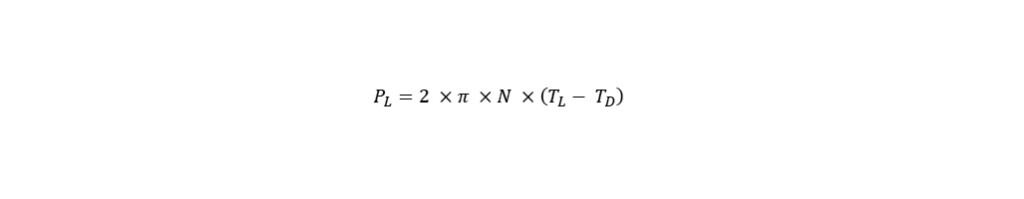
References:
Asnaghi, M., Smith, T., Martin, I., & Wendt, D. (2014). Bioreactors. https://doi.org/10.1016/B978-0-12-420145-3.00012-2
Bylund, F., Collet, E., Enfors, S. O., & Larsson, G. (1998). Substrate gradient formation in the large-scale bioreactor lowers cell yield and increases by-product formation. Bioprocess Engineering, 18(3), 171–180. https://doi.org/10.1007/s004490050427
Croughan, M. S., Hamel, J. F., & Wang, D. I. C. (1987). Hydrodynamic effects on animal cells grown in microcarrier cultures. Biotechnology and Bioengineering, 95(2), 295–305. https://doi.org/10.1002/bit.21158
Gray, D. R., Chen, S., Howarth, W., Inlow, D., & Maiorella, B. L. (1996). CO2 in large-scale and high-density CHO cell perfusion culture. Cytotechnology, 22(1–3), 65–78. https://doi.org/10.1007/BF00353925
Osman, J. J., Birch, J., & Varley, J. (2001). The response of GS-NS0 myeloma cells to pH shifts and pH perturbations. Biotechnology and Bioengineering, 75(1), 63–73. https://doi.org/10.1002/bit.1165
Xing, Z., Kenty, B. M., Li, Z. J., & Lee, S. S. (2009). Scale-up analysis for a CHO cell culture process in large-scale bioreactors. Biotechnology and Bioengineering, 103(4), 733–746. https://doi.org/10.1002/bit.22287


Khushbu Kumari
According to Value Market Research, the latest technology trends and global market opportunity analysis in the Cell Culture Media Market industry growing with a high CAGR in the upcoming year. Our report has categorized the market based on technology, service, development, vertical and region. https://www.valuemarketresearch.com/report/cell-culture-media-market
Vivek Srivastava
Very interesting indeed, I really liked the step by step queries and their explanations.
A good starting place for a new bee to understand fermentation and scale up.
Julia Brück
Hello Vivek,
thanks for your feedback. I'm happy that our articles were helpful for you.
Best regards
Julia
Melika Hadjebi
I would like to get more information about cell culture scale up
Julia Brück
Hi Melika,
thanks for reaching out to us. I will contact you .
Best regards
Kulwant
Thomas Sattler
Could you send this white paper please?
Chantal Fiechter Schaub
Dear Thomans, you can download the white paper directly in the blog-post.
Lucas Schimmelpfenning
Really nice work Lea and Kulwant. This is a great informational tool, I appreciate your work on this. We need to get some of the Fed Batch bioreactor data though!
Kulwant Kandra
Thanks Lucas. Yes definitely, we will be working on this.