In order to understand the optimum conditions for in-vitro culture, you need to know which natural environment gives the organism what they need to grow and thrive. In addition, a clear understanding of the different growth phases in a bioprocess is essential for selecting a bioprocess strategy.

In this blog post, we’re going to focus on the key aspects that are important for understanding and defining a bioprocess.
Choosing the perfect cultivation conditions
Practically all organisms of eukaryotic and prokaryotic origin (i.e., with or without a nucleus) can be cultured in an incubator shaker or in a bioreactor. In order to understand the optimum conditions for growth, you need to know what natural environment provides an organism with all it needs to grow and thrive. A short overview of standard cultivation parameters for commonly used microorganisms and cell cultures is summarized below.
Bacteria
Bacteria cover a wide range of possible growth conditions. In addition to “normal” bacteria, there are also those that love extreme environmental conditions, the so-called extremophiles. Examples of these include thermophilic (which like high temperatures) and halophilic (which like high salt concentrations) microorganisms. The doubling time, i.e., the time needed until a microorganism population has doubled, can vary from minutes to days. This can depend, for example, on whether it is anaerobic or even genetically modified bacterium.
Typical cultivation parameters for bacteria:
| Parameter | Temperature | Mixing rate | Cultivation time | pH | pO2 | |
| Shaker | 20 – 60 °C | 100 – 400 min–1 | 8 – 60 h | |||
| Bioreactor | 20 – 60 °C | 100 – 1500 min–1 | 8 – 60 h | 7.0 | 0 – 80% | |
Yeasts
Yeasts have the same essential mixing, temperature, etc., requirements as bacteria. That’s why these do not normally require any special conditions for growth – they typically prefer a process temperature of 30 °C and an acidic pH. Good mixing and an adequate supply of oxygen are essential for a good yield of yeast biomass. Yeast metabolism generates a great deal of heat, and you may need to cool the process in order to counteract this and maintain an acceptable operating temperature.
Typical cultivation parameters for yeasts:
| Parameter | Temperature | Mixing rate | Cultivation time | pH | pO2 | |
| Shaker | 25 – 30 °C | 200 – 250 min–1 | 16 h | |||
| Bioreactor | 25 °C | 1000 min–1 | 48 h | 6.5 | 0 – 50% | |
Fungi
Fungi often grow as filamentous, mycelial cell aggregates that are often sensitive to shear. This is why they tend to adhere to the vessel wall or even in the headspace – in order to escape the shear stress. The mycelia form linear strands or pellets when stirred . This can result in a very viscous culture medium requiring considerable power input for mixing and gassing. This often makes it tricky to identify ideal operating parameters for cultivating fungi.
Typical cultivation parameters for fungi:
| Parameter | Temperature | Mixing rate | Cultivation time | pH | pO2 | |
| Shaker | 23 °C | 250 min–1 | 72 – 90 h | |||
| Bioreactor | 23 °C | 1500 min–1 | 8 – 72 h | 5.0 – 6.0 | 25 – 50% | |
Cell cultures
Cultivating plant or animal cells involves obtaining a primary culture of cells, tissues or organs that possess full metabolic capacity. Further subculture in an incubator or shaker produces cell lines capable of repeated cell divisions over a longer time, which can then be further cultured in the bioreactor. Within cell cultures, a distinction is drawn between adherent cells (growing on a solid surface) and suspension cultures (floating in the culture medium).
Typical cultivation parameters for mammalian cells:
| Parameter | Temperature | Mixing rate | Cultivation time | pH | pO2 | |
| Shaker | 37 °C | 40 – 120 min–1 | 96 h+ | |||
| Bioreactor | 37 °C | 40 – 150 min–1 | 7 – 70 days | 7.2 | 30 – 50% | |
For an overview on cultivation parameters for insect cells and plant cells have a look at our blog post: Typical Cultivation Parameters for Cell Culture.
The 4 basic phases of a bioprocess
The objective of a bioprocess is to use cells or their components to generate more of a “product” in the most efficient way possible. This can be the biomass itself or components of cells which then require further processing. This downstream processing usually requires a great deal of effort. In order to select a suitable bioprocess strategy, a fundamental knowledge of the different phases of a bioprocess and their characteristics is essential.
1. The Lag phase
Following the inoculation of a culture in the bioreactor, cells first have to adjust to their new environmental conditions. This period is called the latency or lag phase. Although there are sufficient nutrients and, most importantly, no metabolic waste products in the fresh medium, the microorganisms/cells still can’t grow at full speed because they haven’t yet adapted to the environment. They adapt by sensing environmental parameters such as temperature and nutrient supply in order to regulate the appropriate genes. This can take some time, depending on the organism.
Tip: One way to shorten the lag phase is by starting a preculture in the shake flask under the same conditions (same medium, same temperature) and rapidly transferring the cells to the bioreactor during the exponential growth phase.

Schematic representation of the lag phase.
The inoculum added to the culture medium defines the starting quantity of cells. After the bioreactor is inoculated, the viable cell count initially increases slowly, because the organisms must still adapt themselves to the prevailing environmental conditions. This phase is therefore called the latency or lag phase.
2. The Log phase
In the subsequent exponential phase, called the log phase, the microorganism is ideally adapted to the environment, and the growth rate of the dividing cells is at its maximum. The term “exponential growth” stems from the fact that the number of cells does not increase linearly—it doubles constantly. In other words, it is not just 2, 3, 4, or 5 cells that appear, but rather 4, 8, 16, 32 cells, etc.
The cells divide at the greatest possible speed, and the biomass grows. Now the nutrients are absorbed and metabolized at the maximum rate, which, in an aerobic bioprocess, increases oxygen requirements and carbon dioxide production. In short, the nutrients are used up as quickly as possible. During this process, the bacteria also produce by-products such as organic acids or excess heat. When this happens, researchers need to draw upon the bioreactor’s broad range of tools in order to prevent cell growth from being impaired. In the log phase, the increasing amount of biomass and any lysed (disrupted) cells increases the content of free proteins in the medium and the risk of foam formation.
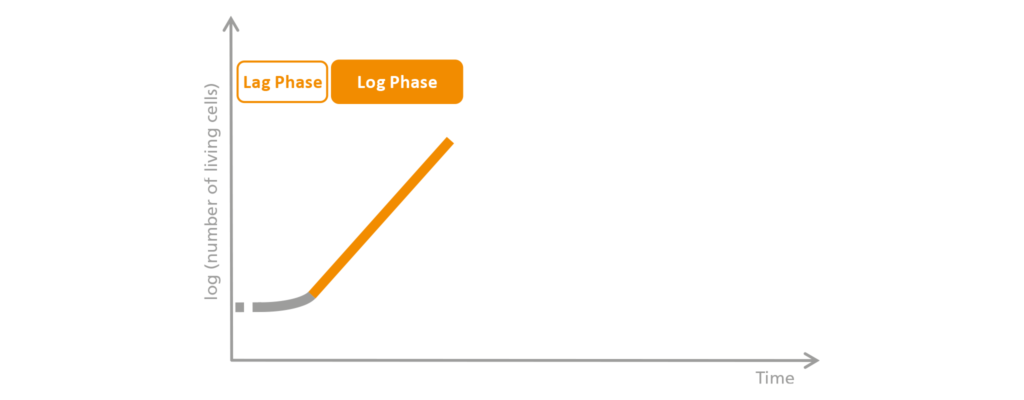
Schematic representation of the log phase.
The cells are now well-adapted and divide at the greatest possible speed since they have all the nutrients they need. For this reason, the number of living cells increases rapidly during this phase, which is called the exponential or log phase.
3. The stationary phase
Unfortunately, exponential growth cannot go on forever. Nutrients are depleted and toxic components of dead cells accumulate. The cells are becoming more and more “uncomfortable” in the bioreactor, and thus the growth rate decreases in the subsequent, stationary phase. In this phase, however, the proliferation and death of the microorganisms or cell cultures are still in balance.
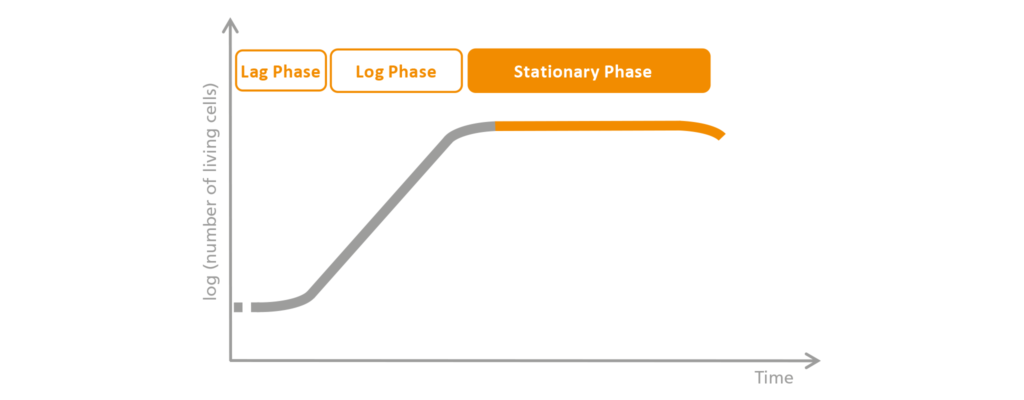
Schematic representation of the stationary phase.
The nutrients are now exhausted and harmful products begin to accumulate. For this reason, the cells begin to divide at a slower rate, to the point that the number of dying cells is approximately equal to the number of cells produced through division. This phase is therefore called the stationary phase.
4. The Death-phase
At the end of the bioprocess, in what is known as the death or die-off phase, the growth rate drops so far that many more cells die than are added by new cell division. This results in a net loss. As a consequence, depending on the process management, either the bioprocess comes to a natural end or the user deliberately stops it. This also depends on how long it makes sense for a process like this to keep going. If the bioprocess is not actively ended, the cells gradually die off because they have simply used up their food and the toxic effect of accumulating metabolites does the rest.
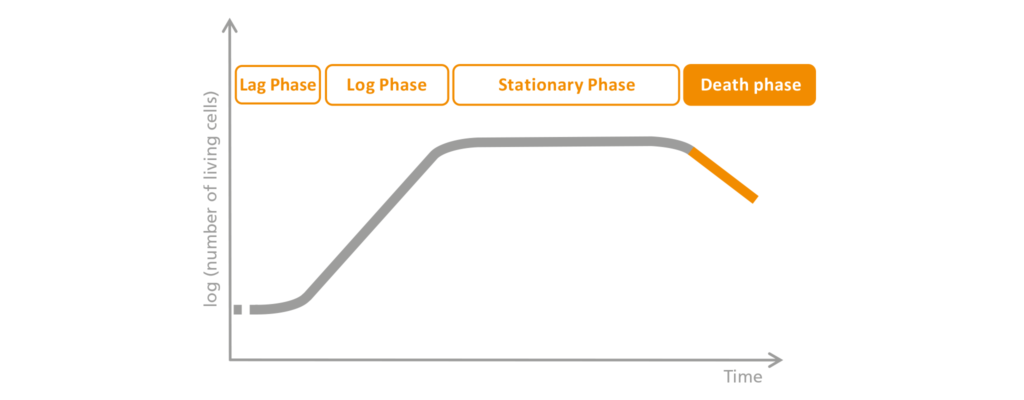
Schematic representation of the death-phase.
If the bioprocess is allowed to advance to the death phase. This phase is characterized by a decreasing number of living cells, due to insufficient nutrients and the accumulation of harmful by-products. Since more cells die off than are added by division, the number of living organisms decreases.
Selecting the right process strategy allows you to optimize the design of a bioprocess, while maximizing efficiency in terms of product yield and time. In order to get an overview on different bioprocess strategies have a look at our blog post: The Difference Between Batch, Fed-batch and Continuous Processes.
Downstream Processing
Once complete, the bioprocess is normally followed by the downstream processing, i.e., harvesting and processing desired products. This means that the fraction that a researcher keeps and processes can vary, depending on the process and the desired end product. Take, for example, a recombinant protein, which, when released by the microorganism or cell line into the medium, can be recovered simply by processing the medium. For a similar protein which cannot be secreted – the scientist first has to separate the protein from the cell (and the complex mixture of lipids, proteins, nucleic acids and sugars). The product characteristics determine whether purification is simple or more elaborate as well as the number of steps. These procedures have implications on yield, time, labour and cost.
Generally, downstream processing stages include:
- Cell separation
- Cell disruption
- Product extraction
- Concentration
- Purification
- Packaging
At the same time, the bioreactor has to be cleaned and prepared for the next process, either manually or following CIP and/or SIP procedures.

Summary:
This overview has outlined three key areas of bioprocessing for a range of organisms, namely:
- The correct culture conditions for the classes of organisms most commonly used in commercial bioprocess, such as food, beverage and pharamceutical manufacture.
- The different phases of growth for any of these organisms broadly follows the same path, while showing great variation in the timing and yields of their respective bioprocessing.
- How the goal of the bioprocess is reflected in the amount of effort required for various downstream steps to obtain a finished product.

Ian Miller
To share with customers.