This article will look at the history and development of a de-facto standard in academic and industrial research laboratories throughput the world, the Multitron incubator shaker. The aim is to show that an initial revolution can lead to evolution, as needs change over time. The fundamentals of a good design can last decades, when new benefits and the best of current technologies are added regularly.

To provide a little context from a historical perspective, the use of orbital shakers for submerged culture in shake flasks dates to the mid 1930’s and concentrated on fungal culture. Key parameters like shaking throw, an orbital movement for good growth and speed range were established early. This made the shake flask useful for seed train production when Stirred Tank Reactors (STR’s) first came into use in the 1940’s for antibiotic production. By the mid 1960’s, the shaker and its temperature-controlled sibling, the incubator shaker, were in common use as a standard laboratory tool for the biosciences. In addition to shake flasks, test tubes and microwell plates were being used with shakers to provide good mixing and oxygen transfer.
1991: Focus on fungal and microbial applications
The Multitron development dates to the 1990’s and came from the need for maximum shake flask capacity for the minimum space in the laboratory. In previous decades, the need for high capacity and throughput was met by free-standing, large shakers housed in constant-temperature rooms. The main application for these units was often large-scale screening for antibiotics. These systems were simple, effective for their main task but extremely inflexible for accommodating several users with different needs e.g., different growth temperatures. The alternative, an incubator shaker was more flexible, but lacked capacity, both in number of flasks and their maximum size.
The first Multitron provided a “game changer” in that it made use of vertical space in the laboratory by stacking up to three incubation chambers. This provided its own design challenges to keep the top deck accessible, providing easy flask handling, prevent any spillages from moving down the stack, and allowing easy access for cleaning and servicing.

This system was a genuine revolution and met the changing needs of researchers engaged in microbial work. However, applications were changing with the rise of cell culture for mammalian or insect cell lines.
2008-2011: Cell culture applications come to the fore
By the early 2000’s, the need for growth of mammalian and insect cells in suspension culture was clear. Microbial and fungi/yeast hosts for recombinant protein production were limited in their ability to provide complete glycosylation. Also, production of viral vectors for vaccines and recombinant work using both insect and mammalian cells were common.
This led to a new generation of Multitron with a larger 50 mm shaking throw for gentle mixing plus the ability to measure and control parameters such as gassing with CO2 and percentage relative humidity. The excellent temperature distribution, a certified antimicrobial surface and options such as darkening for insect cells infected with Bacculovirus made the Mulitron the “ideal home” for these cultures.

2012-2017: Multiple, parallel experiments and green applications
Then, things changed and applications such as algal biofuel and massively parallel screening in microtiter plates came to the fore. The need for thousands of screening experiments to provide rapid screening for specific biological activity drove an increased use in 96-well plates. They needed special options and adaptation to accommodate large numbers per chamber and to stop wells drying out over time. Providing adequate mixing for oxygen transfer was an issue in the smaller wells. A special high speed, short throw drive system solved this problem.
Next to the “classical” technical shaker features, influencing culture growth and productivity, usability topics have become more popular from the mid 2010’s onwards. Serial communications gave way to networked connectivity to bioprocess software for logging, analysis, and validation of shaker-based processes. An incubator shaker which could be qualified like a bioreactor for such validated process was a real revolution this time around. Suddenly, the results of screening could be taken to small scale clinical trials with the output from a single, stacked system.
When biofuel applications using photosynthetic bacteria and algae to produce fermentable sugars, the Multitron evolved to meet these requirements, yet again. LED lighting to encouraged growth of phototrophic cell lines with special spectra.
2019: Digitalization & Performance – The users in the 2020’s want even more
Once more, things have changed again and the modern bioprocess in incubator shakers is demanding even more information from the shake flask and better performance from the incubator shaker. Some options for increasing the amount of measured data and control have been available previously, but the need for automation and efficiency at the research scale is driving more users towards taking up these solutions. Data integration from different sources is becoming vital for screening, validation, and optimization purposes. Two different options are available, depending on the preferred platform: The INFORS HT software package eve® is able to plan, control and analyze your bioprocesses. The software integrates workflows, devices, bioprocess information and big data in a web-based platform, with which you can organize your bioprocesses in a web based system. Alternatively, those customers who are using an industrial SCADA system for organizing the data from the whole facility, can connect their incubator shakers to the SCADA via Profibus DP, MODBUS TCP gateways or the tried-and-tested analogue outputs.
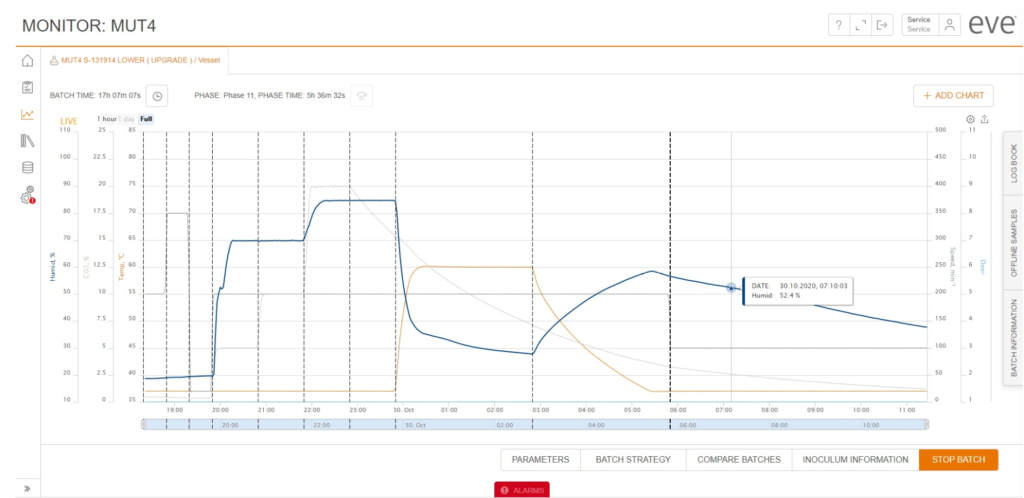
Chart overview of incubation shaker data using the eve bioprocess platform software with parameters temperature, humidity, shaker speed, door status (open/closed) and process phase changes. In addition batch and phase time as well as current alarms are displayed.
Another key performance improvement required by many users relates to the standardization of mammalian cell culture for many bioprocesses, especially CHO cells. The requirement for carbon dioxide gassing is mandatory for growth. However, the design of incubator shakers makes gas throughput higher than for incubators alone. Taking this to the minimum possible can save both costs and labor. The Multitron is designed to be as gas tight as possible, while still providing sufficient aeration for oxygen.
2023: A new display and new humidification system offering additional functionality
While retaining benchmarks of the previous display like excellent readability from a distance, the new display technology comes with additional functionality: the Event list informs about the 18 last events, like parameter changes, door opening etc. The timer programming table on a single screen eliminates the needs to scroll different pages. The self-explanatory messages provide clear information about the current status. The on-line help menu guides the user through the settings. The new HHC Hygienic Humidification Control offers bi-directional control, enabling not only humidification but de-humidification as well. The user benefits from enhanced control precision.
Conclusion: Constant development is the key to sustainable success
These examples show how revolutionary development in design, combined with evolving modern technologies, can meet changing needs. This approach is where the path to the future lies. Experts believe that in the future the incubator shaker will be seamlessly integrated in other lab processes. Better integration can only cement the place of the incubator shaker as a key tool for any bioscience laboratory that needs cultivation capability at a small scale.
Innovations in the fields of a laboratory automation and bioprocess modelling will lead to greater efficiency, optimized productivity, and an enhanced ability to quickly “pick winners”. These changes are coming into use for production environments and the ever-increasing cost of new product development will drive their use for industrial (and academic) research.
The evolutionary approach to Multitron design will continue to provide revolutions for the user by building on what is familiar and good with the latest technology. Here’s to the next 30 years.

Ergonomic design has been a key aspect of all Multitron developments. The downwards opening door with the pull-out tray for easy access to individual flask is still one of the best loved Multitron features from our users.


Antonio Pires
Always a pleasure to read your posts, Tony. Thank you for sharing with us.
Tony Allman
Thanks for the kind words, it's always en encouragement to have good feedback.
Tony.