The shake flask is the standard in many laboratories for cultivation of cells and microorganisms. Thereby achieving high biomass concentrations remains a constant challenge. We show you 5 tricks – you can also watch the webinar at the end of the post!

Low biomass levels can be attributed to:
- Oxygen limitation
- Lack of nutrients
- Cultivation interruptions and repeated handling
This post explains various ways how to increase the quantity of biomass produced in your shake flasks by mitigating the causes of low growth and productivity. Individual experiments may only use some of these solutions, however, there is enough variety covered below to assist with a broad range of applications.
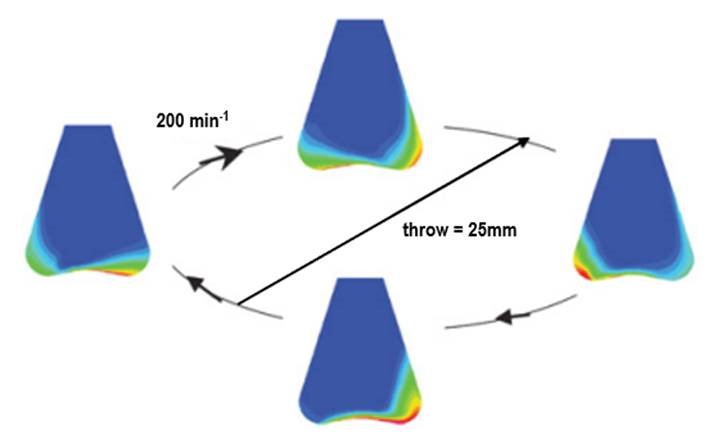
1. Choose the right shaking throw & speed for your flask
Often the reason for poor biomass concentration is not the clone, as you may think, but an external factor.
Two factors that have a big influence on the oxygen transfer and, therefore, the biomass growth is the shaking throw in combination with the shaking speed.
As a general starting point, 25 mm is always a good choice and can yield excellent results independent of the application. However, in some cases, restrictions in respect of oxygen transfer/cell growth will occur and a better result can be obtained if the shaking throw is selected according to the application.
As a rule of thumb:
- 25 mm throw: recommended for flask size from 25 mL up to 2 L
- 50 mm throw: recommended for flask size from 2 L to 5 L
Microtiter- and deep well plates:
- Use 3 mm throw with shaking speeds of min. 800, up to 1000 min-1
Example of suitable settings for a microbial application (Bacteria, Yeasts, Funghi)
- Mostly 25 mm shaking diameter
- Shaking speed: 120 – 400 min-1, mainly around 300 min-1
2. Keep volumes low to improve oxygen transfer
Oxygen transfer in shake flasks is much lower compared to a bioreactor and may represent a limiting factor in shaken bacterial and fungal cultures. A typical shake flask will be filled to 20 – 25 % of its working volume. This is a compromise which can severely limit oxygen transfer into the culture and delay cell growth. It may be enough for processes that just aim to generate biomass or for certain cultures such as micro-aerophilic or slow-growing organisms. If, however the target is to have no limitation at all, it is vitally important to ensure a maximum liquid/gas interface area to allow for O2 and CO2 exchange in the culture.
Reduce the working volume to 10 % of the total shake flask volume. This will “smear” the whole culture in a thin layer on the sides of the flask. This increase of surface to volume ration allows for a higher oxygen transfer rate.
3. Use specialised flask geometries to get better results
The conventional Erlenmeyer shake flask is distinguished by having smooth sides. It was invented in the 1800’s but not specifically as a culture vessel. It allows for an even mixing action but can limit oxygen transfer. Other designs of flask, such as the Fernbach, have baffles that do the same as their counterparts in bioreactors – disrupting the smooth flow of the liquid culture. This creates turbulent areas where gas transfer is improved, and the flasks can even be used with animal cell culture growth.
The Thomson Optimum GrowthTM flasks are even more specialised vessels. They have a unique design including baffles and are suitable for mammalian, insect and microbial cells. Working volumes of up to 60 % of the flask size can be used, increasing the productivity in the incubator shaker. To be able to benefit from such alternative shaking flasks that increase biomass concentration a shaker that is flexible and powerful enough to hold such flasks is needed.

4. Provide enough nutrients
Improving the oxygen transfer alone will not be enough to increase cell growth, as this depends on various factors. If e.g. the carbon source is depleted, the cells will not reach a higher density, no matter how much oxygen is available. So, a lack of care in medium and supplements choice will also lower the biomass concentration. Recommended media composition:
- Substrate / carbon source
- Nitrogen source
- (Additional) trace elements and vitamins
- Buffer for an ideal pH
Since cell growth is inhibited by too high substrate concentrations, it is not a good idea to put in huge amounts of substrate (e.g. glucose) in the media at the start. In consequence, the cells will have taken up all the nutrients provided in the media at some point and therefore stop growing. This can be prevented by feeding the cells in certain time intervals with feed media.
Feeding can be either done manually or alternatively automatically, e.g. using the aquila biolabs LIS (Liquid Injection System). This streamlines the system and opens a range of fed-batch and continuous process possibilities without the manual applications and cultivation interruptions.
5. Minimize interruption: handling and online biomass measurement
Every time the incubator shaker door is opened to access a flask, heat is lost, and the shaking action is halted. Doing this occasionally will not make much difference. However, you should avoid repeated door opening or taking the flask out for a longer time. This could cause a breach in the normal growth curve, introducing uncertainty into your process. Several things can mitigate these effects:
- rapid stopping and re-starting to minimize the downtime for shaking
- fast return to setpoint after interruption through door opening
- automatic feeding
- online biomass measurement
Shake flasks alone provide no online monitoring information, and gleaning data from them requires frequent removal from the growth environment. This disruption most certainly skews results and hinders optimal growth. Human error is inevitably introduced into the process via mistakes in analysis or missed readings.
Non-invasive, accurate and continuous measurements of biomass concentration are possible, e.g. with the aquila biolabs CGQ (Cell Growth Quantifier). It allows to measure multiple flasks at the same time.

Last, but not least, some tips concerning the inoculum:
The output from any culture process can only be as good as the input, any of the below will contribute to poor growth:
- Poor treatment of cells before inoculation
- First use or minimal characterization of the clone
The cell line must be matched with the correct media type and able to be cultivated in shake flasks e.g. no shear limitations. It must be freshly prepared from pre-cultures still in the logarithmic phase of growth. This will minimize lag time and give the cells the best chance to grow quickly.

Summary:
This post suggests ways in which the growth of cultures in a shake flask can be optimised. The key methods mentioned are:
- Choose the right shaking throw and speed for your flask
- Keep volumes low to improve oxygen transfer
- Use specialised flask geometries to get better results
- Provide enough nutrients
- Minimize interruption: handling and online biomass measurement
- Make sure the inoculum is cultivated with care

Moti Rebhun
I am interested in fermentation technology.
Thanks.
,Moti Rebhun
Any info concerning fermentation will be very interesting.
Thanks
stanley han
It's good for me.
Thanks a lot.
Julio Cesar Bedoya
I would like to know if there is a webinar for the system Eve. I would like to see it in action and what could be done with it
Thanks for the 5 way to increase biomass in flaks