Bioprocess development usually starts with screening for the most promising clones or culture conditions. All subsequent steps via process characterization, optimization, and finally, production are building on these first insights. Therefore, it is vital that the data generated during screening is of top quality.

Despite of the importance of the data they generate, incubator shakers are often seen as simple workhorses. Many users think of them the same way they do of a fridge or of an oven. However, it definitely pays to understand which factors influence the quality of an incubator shaker – and how building your process on inaccurate or incorrect screening results can cost a lot of time and money.
This blog post provides five examples of how the right equipment will help you achieve better results and avoid picking the wrong candidates:
- Apples or oranges? Why temperature uniformity matters.
- Why so salty? Prevent evaporation with active humidification.
- Don’t stop shaking: Drive capabilities, online monitoring
- Track what’s going on: Data logging
- Minimize downtimes: Reliability, ease of cleaning
1. Apples or oranges? Why temperature uniformity matters.
Incubator shakers are commonly used for the first screening steps. Which clone will produce the desired product? Which cell line has the best vitality? Which components of the culture media have a positive effect on the product yield, and which are inhibitory? No matter the task, the results should allow for a clear ranking based on the questions asked, e.g., productivity, and not be disturbed by external factors.
As many chemical and biological processes have a strong temperature dependence, the temperature is one of the most important environmental factors for a screening. The temperature is therefore controlled in the incubator shaker and most devices on the market will feature an accurate temperature sensor and good overall control stability. One question is, however, often forgotten: Is the temperature uniform throughout the incubation chamber or is it only accurate where the temperature sensor is placed?
Surprisingly, this is not easy to achieve. There may be cold spots in the shaker where there are connections to the outside world. Other components like the drive could act as a heat source. Finally, the layout of the fans, heater, and cooling components need to be optimized for an ideal air flow pattern throughout the shaker.
Suppliers of high-quality incubator shakers will therefore not only specify a general temperature measurement and control accuracy, but also make a statement about temperature uniformity and offer supporting documentation as part of their validation package.
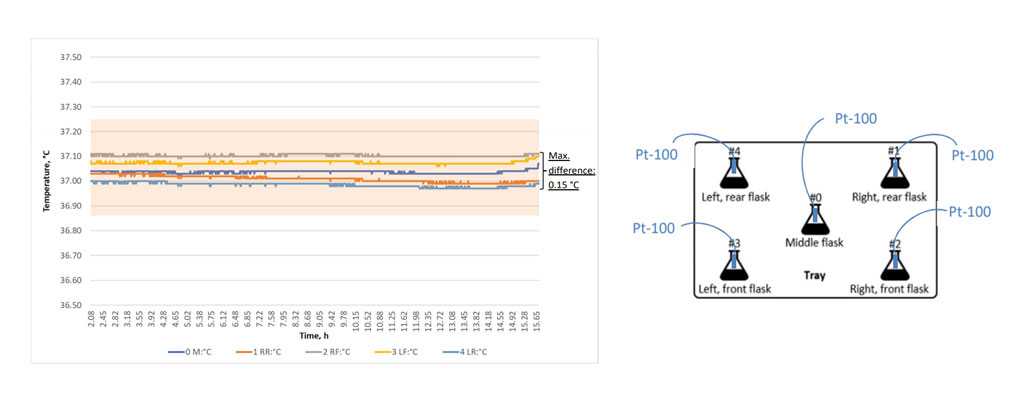
Left: Example data from a temperature uniformity validation showing the uniform temperature control of an INFORS HT Multitron incubator shaker. Right: Excerpt of a protocol for temperature uniformity validation.
2. Why so salty? Prevent evaporation with active humidification.
One phenomenon no one wants to see in their bioprocesses is evaporation as it is associated with many negative side effects. Even if it doesn’t lead to a complete loss of culture volume for subsequent analysis or the death of the culture it is to be avoided at all cost.
Let’s assume 20 % of the culture volume is lost by evaporation. This corresponds to all media components to gradually becoming 25 % more concentrated compared to the start of the experiment. Did the cells only grow in the beginning before the media got too salty? Or did they become more active once a certain concentration of one media component was reached? Or is the final product concentration only a bit higher because of evaporation effects? The screening results won’t be as valid as they should be.
But why did evaporation occur in the first place? Most bioprocesses require oxygen. It is usually provided using air, e.g., by blowing gas through the culture media in a bioreactor. Unfortunately, this not only brings oxygen into the media and remove exit gases like carbon dioxide, but also causes a portion of the culture media to evaporate and get carried away with the exit gas stream. Bioreactors typically feature exit gas coolers to turn the escaping water into condensate which will then drip back down into the culture vessel.
An incubator shaker doesn’t provide active gassing into the culture media. The shaking motion swirls the media in the culture vessels, effectively increasing the overall surface area available for gas transfer. This comes with the same side effects as sparging in a bioreactor and some of the liquid will evaporate over time. It is not that easy to catch the escaping water from shake flasks as they lack a device to do this, such as the exit gas cooler used in a bioreactor.
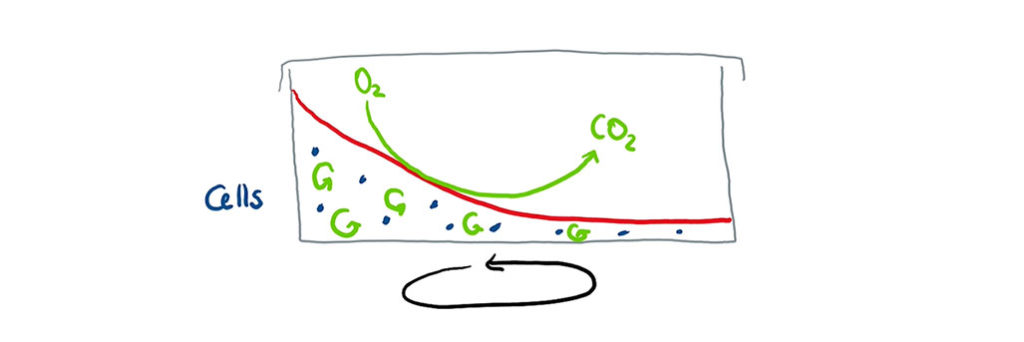
Liquid and cells in a shaken culture vessel.
The solution is to avoid evaporation in the first place by humidifying the atmosphere inside the incubator shaker. If vaporized water is already present, the evaporation rate from the cultures will reduce drastically. Humidification systems should feature precise control of the relative humidity to provide good reproducibility. Hygiene should also be considered as a factor (see factor 5 below). Both requirements can be met with ease by using humidification systems based on an active feed of clean steam. Systems using passive water reservoirs can struggle to meet these criteria.
3. Don’t stop shaking: Drive capabilities, online monitoring
The main function of an incubator shaker is to keep the culture in motion. Whenever that motion stops, e.g., because the researcher opened the door, the cultures are no longer mixed, the cells might sediment and the oxygen transfer rate suffer a large drop. The take home message is: don’t stop shaking!
If a user wants to add new cultures to the shaker or remove finished experiments, there is no way to avoid opening the door. However, the interruption can be kept to a minimum if the shaker has a convenient operating height, the door is easy to operate, and the drive automatically stops and starts instantly.
Operators will commonly stop the shaker to take out cultures for sampling. This will not only cause an interruption for all cultures in the chamber, but the flasks or plates being sampled will often be taken out of the shaker and sit under a hood for many minutes. The negative effects will multiply: not only are mixing and oxygen supply compromised – the surrounding atmosphere including temperature, humidity and carbon dioxide concentration will vary as well.

Online measurement in shake flasks with the cell growth quantifier (CGQ) from aquila biolabs.
While some analyses may require samples to be taken, the sampling interval can be greatly reduced by using online sensors, e.g., the aquila biolabs CGQ (Cell Growth Quantifier). It can be placed under shake flasks and will record the change of the biomass concentration in the cultures over time using backscattering. The user will have to interrupt shaking less often and even gains additional insights into what happens between samples. This makes the screening results more meaningful.
Finally, some processes require liquids, e.g., an inducer, to be added to the cultures at a certain point. This task is also traditionally performed under a a clean hood or biosafety cabinet, with the shake flasks being removed from their optimum environment. Devices like the aquila biolabs LIS (Liquid Injection System) can be used to automate the liquid addition within the shaker, eliminating the need to interrupt shaking.
4. Track what’s going on: Data logging
The demand for more data has been an issue in many areas of bioprocessing for decades. More sensors are added to processes, information about media compositions is stored together with bioprocess data and the “history” of the precultures are just some examples of the increase in process information. However, datasets for screening in shakers often consist of only one data point at the end of the incubation period.
The lack of insights into what happened in the culture can be resolved by more frequent sampling or, to avoid the side-effects of this, by adding online monitoring for shake flasks inside the incubation chamber. Ideally, the information from the sensors can be read by a central software that also stores the data from other bioprocesses, e.g., from bioreactors. This will give researchers a complete overview of all information starting from screening through process optimization up to the final production runs.
However, it’s not just the data from within the shake flasks that matters. The conditions inside the shaker can have a significant effect on the performance of the individual cultures as well. How often did the shaking motion stop? Was the temperature controlled precisely and how uniform was it? Was the humidification turned on at all times? High end shakers can output all these process parameters using a digital communication protocol. Ideally, this would be Ethernet-based so that the incubator shakers can easily be integrated into an existing laboratory network and accessed by the SCADA software.
If information generated by the shaker itself is not enough, extra sensors can be added, e.g. for an external reference measurement of the temperature. Sealed and well insulated cable passthroughs are available to avoid the extra sensors interfering with the conditions inside the incubator chamber.
5. Minimize downtimes: reliability, ease of cleaning
If you have an incubator shaker that fulfils all the requirements to deliver valid screening results you definitely want to get the most out of it. Ideally, it will be the workhorse in your lab that runs 24/7. Ask your supplier what life expectancy the critical components like drive system and circulation fans are designed for. That is a key indicator of how long you can expect to work with the system, and how likely it is to suffer from an unexpected breakdown.
Even the best systems will require periodic inspection to ensure performance remains consistent. Therefore, it is recommended to plan for regular preventive maintenance, typically on an annual basis. A good service agreement will specify that certified personnel will replace all worn parts and inspect all critical components for unusual wear. This ensures the equipment should run without issues until the next inspection. If the device is designed with accessibility and serviceability in mind, the downtime for maintenance tasks can be reliably planned and kept to a minimum.

Clean and hygienic design of a Multitron incubator shaker.
To minimize the chance of unexpected nasty surprises such as contaminations it is recommended that operators clean their incubator shakers regularly. With a well-designed machine, there will be very few dirt traps or difficult to reach spots in the incubator chamber. Corners should be rounded off and complex components of the drive system should be located on the outside of the chamber. Imagine a shake flask breaking and not being able to clean and resume operations yourself, but having to wait for a certified service technician! With a well-designed system the time required for cleaning and the risk for contaminations between cleaning cycles can be kept at a minimum to reduce downtime.
Summary
Screening is a vital first step in bioprocess development and many key decisions will be based on the screening results. The data must therefore be valid and as free from external influences as possible. The most important factors are discussed above and, fortunately, most of them can be minimized or even eliminated. All that is required is taking some extra time and asking the right questions when choosing an incubator shaker to use for your screening, especially regarding key criteria such as:
- Temperature uniformity
- Humidification capabilities
- Minimizing shaking interruptions
- Data logging
- and downtime
